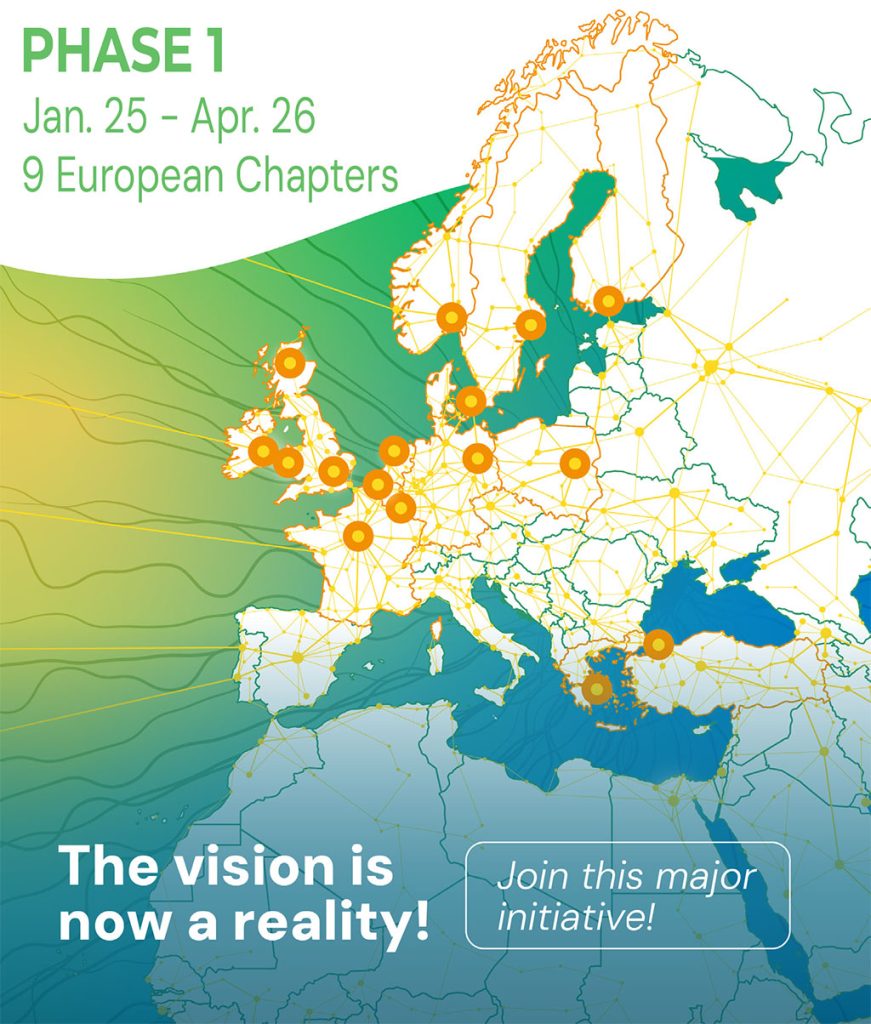
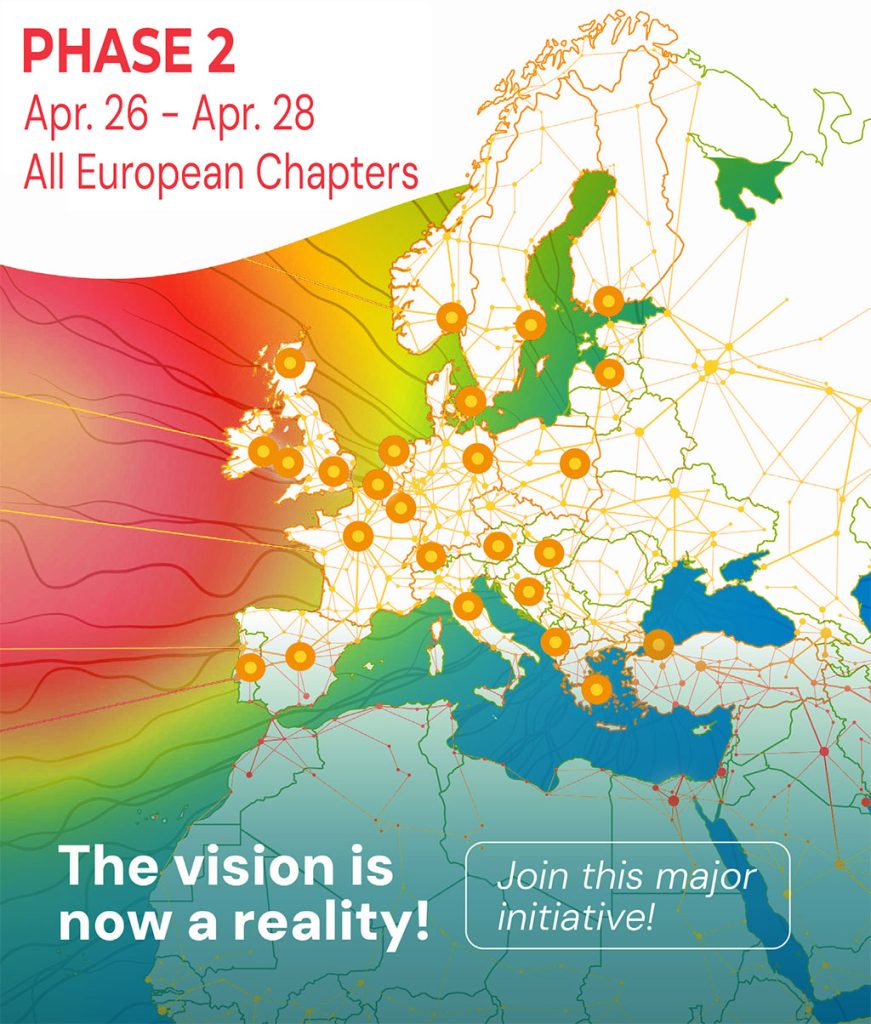
About the INR
Neuromodulation therapies are increasingly used to treat chronic neurological and pain conditions, yet the evidence base remains fragmented, heterogeneous, and difficult to generalize across healthcare systems.
The International Neuromodulation Registry (INR) is a global, multi-stakeholder data platform led by the International Neuromodulation Society (INS) that was established to address these limitations by providing a standardized, multinational platform for real-world evidence generation.
INS Mission Statement
The Mission of the INS is to promote and disseminate the science, education, practice and accessibility of all aspects of neuromodulation. This multidisciplinary society believes that all scientists, doctors, bioengineers, professions allied to medicine and industry partners who have a specialist interest in neuromodulation can work with this society to share science and encourage best practice for the good of humanity.
The Vision of the INS is to harness all scientific, clinical and engineering endeavour throughout the world and to brand neuromodulation to encompass all neurological technologies that through electrical or chemical means improve the function of the impaired individual.
INR Purpose & Impact
The INR is designed to systematically capture high-quality, real-world clinical, procedural, and patient-reported outcomes data across neuromodulation therapies, enabling robust evidence generation to inform clinical practice, regulatory decision-making, and innovation.
Neuromodulation is a rapidly evolving field with expanding indications and technologies; however, there remains a critical need for longitudinal, standardised, real-world evidence (RWE) to complement clinical trials. The INR addresses this gap by providing a scalable, international infrastructure for prospective data collection across diverse healthcare settings. This includes harmonised data models, consistent outcome definitions, and the integration of patient-reported outcome measures, ensuring that data generated are both scientifically rigorous and clinically meaningful.
The INR is positioned to play a pivotal role in supporting post-market clinical follow-up (PMCF) and lifecycle evidence generation for neuromodulation devices. By generating real-world data on safety, performance, and long-term patient outcomes, the registry can help address evolving regulatory requirements and support value-based healthcare decision-making. In parallel, it provides a neutral, independent platform that fosters collaboration between clinicians, researchers, industry partners, and patient communities. Ultimately, the INR aims to improve patient outcomes by enabling data-driven clinical decision-making, identifying best practices, and accelerating innovation in neuromodulation.
| Purpose | Expected Impact/BenefitPurpose |
|---|---|
| Offer independent evidence of performance and quality assurance | Provide clinicians, hospitals, and national modules with validated data for quality improvement and demonstrating the value of care. |
| Provide ability to assess similarities and differences by geographic regions | Identify regional variations in practice to support the development of globally consistent best practices. |
| Support device recalls and post-market surveillance | Enhance patient safety by facilitating rapid identification and response to device-related issues, leading to better regulatory compliance. |
| Create a diversified pool of anonymized data for research | Accelerate scientific discovery by providing researchers with an unparalleled, real-world international dataset. |
| Develop a model for international collaboration and research | Establish a repeatable framework for global data sharing, fostering a collaborative research community. |
| Refine minimum data specification and data standards | Ensure the collected data is robust and interoperable, guaranteeing the reliability and analytical power of the pooled dataset. |